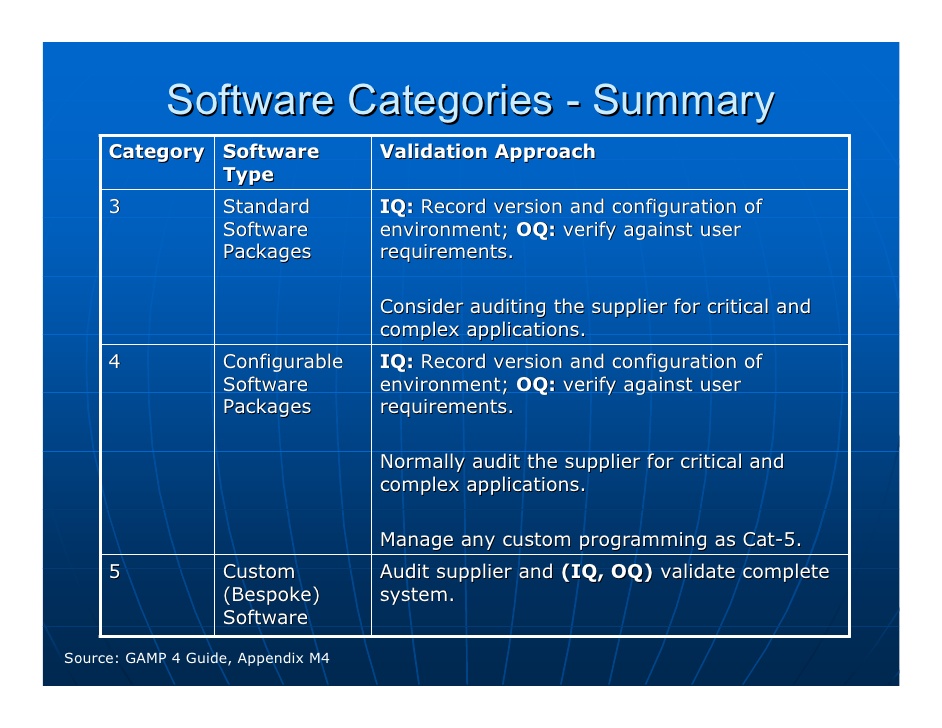
Testing leads to (ideally) Internal Acceptance, Factory Acceptance, and Site Acceptance. The latter two elements form the foundation of a traceability matrix which creates a basis for formal testing. It begins with a User Requirements Specification, which leads to a Functional Requirement and a Design Specification. The guide also outlines a formal process of documentation, testing, and procedure that validate the necessary specifications for the product. It then establishes a system lifecycle approach which covers good practices for the whole production process. First it outlines commonly used terminologies so that everyone can align on the production approach it suggests depending on the category of product. A closer look at the contents of GAMP® 5Īs a whole, the guideline provides an interpretation of regulatory requirements in the field of pharma manufacturing, specifically, about computerized pharmaceutical production systems. For manufacturers, GAMP® 5 guides them to ensure their products meet necessary standards according to a risk-based approach to compliance. In practice, this means that these recommendations apply both to the users of automated pharmaceutical products, as well as the manufacturers who create and market them.įor users, the guideline outlines the principles that they should be aware of which assure computerized pharmaceutical products are appropriate for their intended purpose. GAMP® 5 aims to provide a comprehensive explanation of how pharmaceutical companies should validate their computer systems. Making the Case for ALM in Pharma Project Management Who does it apply to? These best practices are respected and used by regulated companies and their suppliers all over the world. In other words, this guideline helps manufacturers meet regulations they must comply with to go to market. 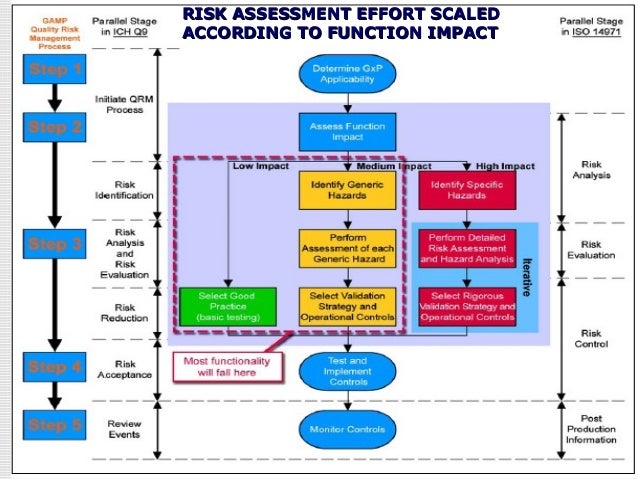
It is important to note that rather than being a regulation, GAMP® 5 is a set of principles and procedures created to help validate automated computer systems for manufactured pharmaceutical products. GAMP® 5 is the latest standard of the guideline it was released in February 2008 by the International Society for Pharmaceutical Engineering, also known as ISPE. In essence, GAMP represents a structured approach to validating computer systems in digital pharmaceutical products. Created in 1991 by pharmaceutical professionals, it was specifically designed to address industry needs and meet the evolving expectations of the FDA and regulatory bodies in Europe for computer system compliance and validation. GAMP - or the Good Automated Manufacturing Practice - is the definitive industry guideline for creating compliant computer systems. To create a computer-based system that serves its purpose in a reliable, transparent, and most importantly safe way, companies developing pharmaceutical products need to meet various predefined requirements. Read on to learn more about who it applies to, its contents, requirements, and systems validation according to GAMP® 5!Ĭomputer-based systems validation in regulated industries is no walk in the park. 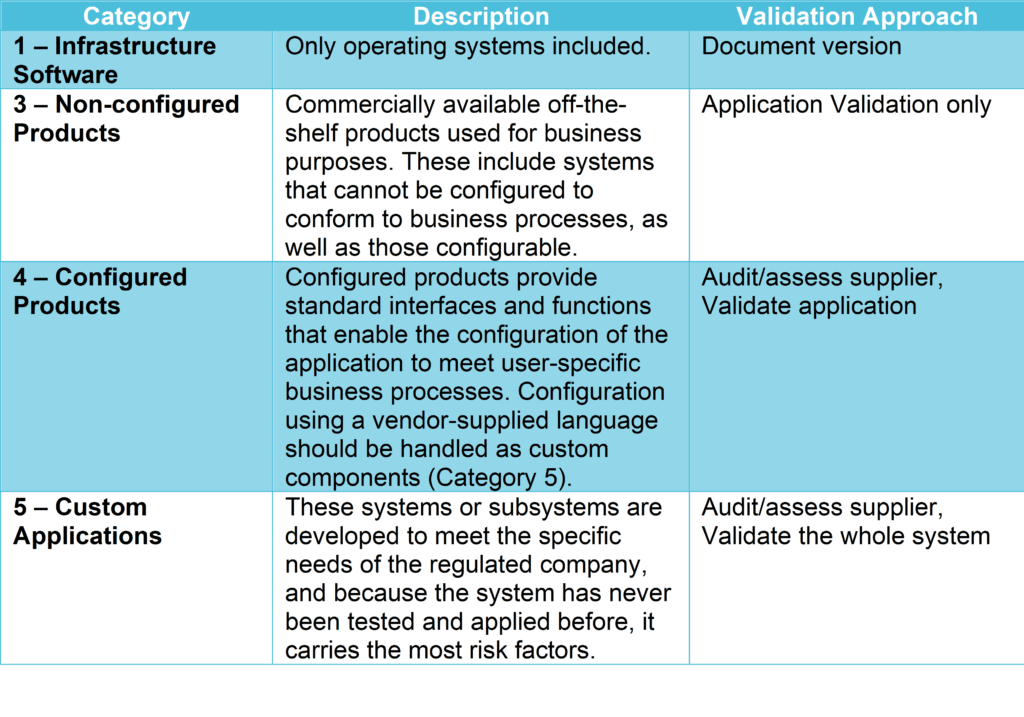
A detailed overview of Good Automated Manufacturing Practice guidelines (GAMP® 5).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
